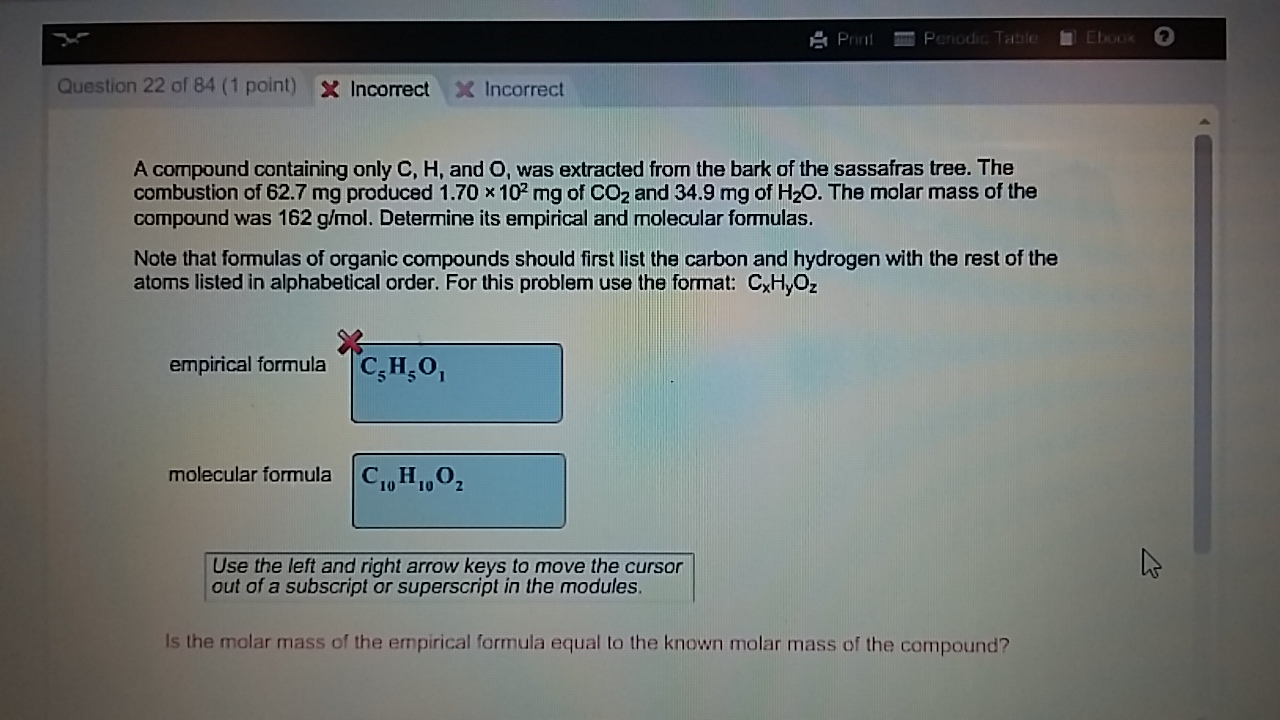 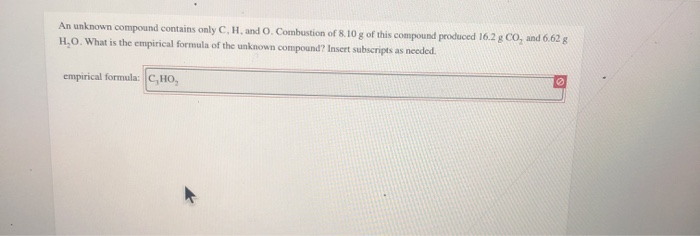
Now we need to convert the moles to grams of these elements 0.006430 mol C xįind the mass of Oxygen by subtracting the C and H from the total mass of the sampleĠ.1005g= 0.07716 g C + 0. An unknown compound contains only the three elements C,H, and O. What is the empirical formula for menthol? 0.2829 g of CO 2 x This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. a) Write a balanced chemical equation for the combustion of methane. Experts answer 1) Determine the grams of carbon in 9.42 g CO 2 and the grams of hydrogen in 2.57 g H 2 O. What is the empirical formula of the unknown compound. 
Combustion of 4.00g of this compound produced 9.42 g of CO2 and 2.57 g of H2O. A 0.1005 g sample of menthol is combusted, producing 0.2829 g of CO 2 and 0.1159 g of H 2O. An unknown compound contains only C, H, and O. Menthol, the substance we can smell in mentholated cough drops, is composed of C, H, and O. Give the balanced equation for each of the following chemical reactions : a.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
